 Due to their highly reactive nature, these elements are commonly stored as an inert fluid material, such as petroleum-derived oil. As a result, the metal reactivity series may be defined as a set of metals grouped in decreasing order of reactivity. With respect to the reactivity of metallic elements listed on the periodic table, the metallic elements are broadly classified into four groups:Īctive metals are characterized by their tendency to readily combine with gaseous oxygen and atmospheric water vapor due to a single electron in its outer shell that can be readily exchanged to form a cation and ultimately lead to a chemical reaction. These elements belong to groups IA and IIA of the periodic table. The most active metals in the activity series are lithium, sodium, rubidium, potassium, cesium, calcium, strontium and barium. The reactivity series is a series of metals, in order of reactivity from highest to lowest. The video shows an experiment to determine the placement of three different metals (Cu, Zn and Mg) on the activity series.All active metals are found in Group I of the periodic table (i.e., on the left side of the periodic table), except hydrogen, which is not considered a metal and lies at the top left hand corner of the periodic table. If we look at the activity series, we see that aluminum is above copper, so this reaction will occur, and the aluminum will replace the copper to form aluminum sulfate. The most chemically reactive metal with a stable isotope is cesium. #"Cu(s) + Zn(NO"_3)_2("aq")"#rarr#"no reaction"#Įxample 2: Will the following single replacement reaction occur? 
If we look at the activity series above, we see that copper is below zinc, so it cannot replace the zinc in the zinc nitrate compound. 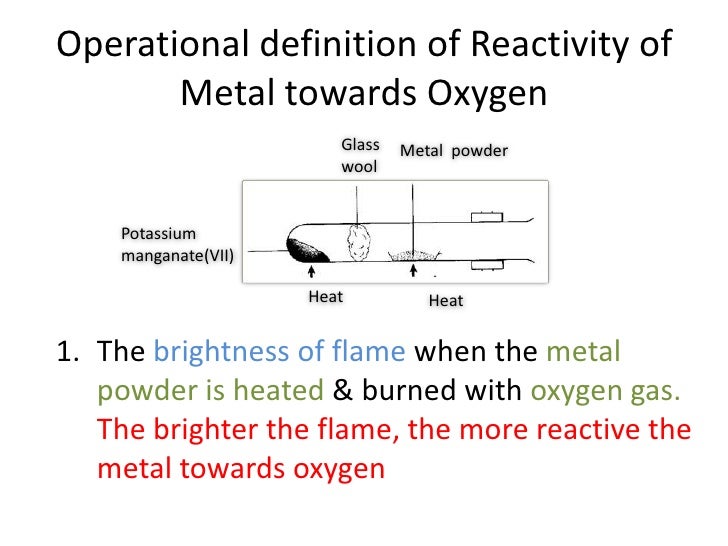
The reactivity series is also known as the activity series of metals. Sometimes it contains hydrogen and carbon. This means that a metal can only replace a metal below it in the series.Įxample 1: Will the following single replacement reaction occur? The reactivity series is an ordering of metals from most reactive to least reactive. The reactivity series is a list of metals, ordered from the most reactive metal to the least reactive metal. An activity series of metals lists metals in descending order of reactivity. 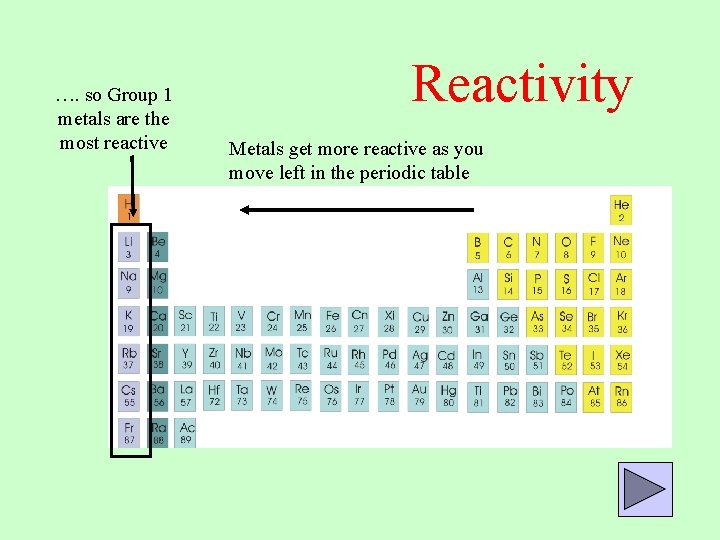
In order to determine whether this will actually happen, you consult an activity series of metals. Where #"A"# and #"B"# are metals, and #"C"# is a negative ion. 
Activity series of some of the more common metals, listed in descending order of reactivity. It is used to determine the products of single displacement reactions, whereby metal A will replace another metal B in a solution if A is higher in the series. In a single replacement reaction, a metal and a metal compound may react so that the metal will replace the metal in the compound, producing the replaced metal and a new compound with the original metal. The reactivity series is a series of metals, in order of reactivity from highest to lowest. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
